Patients are not treated as one average profile
The underlying model can represent variation in age, disease stage, risk, test results, treatment eligibility, event timing, and survival.
Medip applications are custom built for specific disease areas and pathways. Within each application, users adjust assumptions and run an individual patient model to produce transparent, documented analyses for publications, value dossiers, and market access evidence.
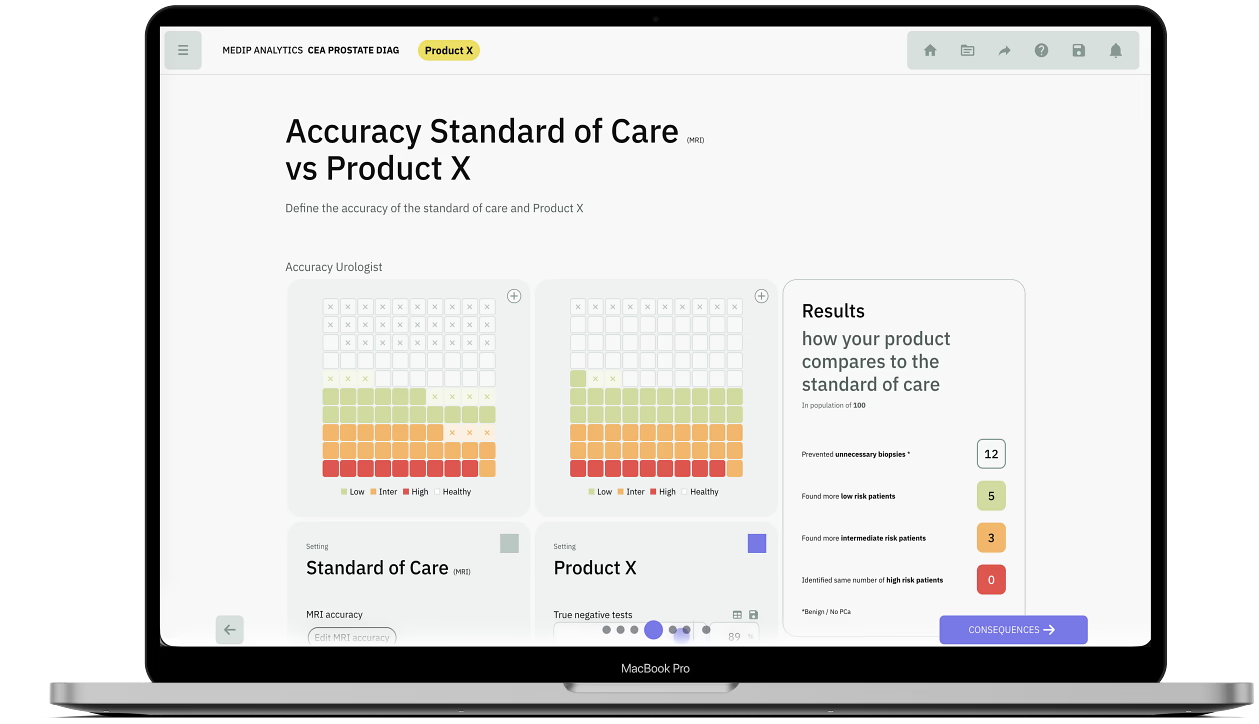
Model patient heterogeneity, event histories, and long-term downstream outcomes.
Support analyses aligned with established assessment expectations such as NICE.
Trace application structure, model assumptions, inputs, analyses, and outputs for review.
Individual patient simulation is useful when outcomes depend on patient characteristics, pathway timing, event history, diagnostic accuracy, treatment sequencing, or long-term disease progression.
The underlying model can represent variation in age, disease stage, risk, test results, treatment eligibility, event timing, and survival.
After users select inputs in the application, the model can follow patients through diagnosis, treatment, monitoring, progression, complications, recurrence, death, and accumulated costs and QALYs.
Diagnostic sequences, treatment choices, follow-up intensity, referral routes, false positives, false negatives, and downstream decisions can be represented explicitly.
Scenario analysis, deterministic sensitivity analysis, probabilistic sensitivity analysis, and threshold analysis can show how robust conclusions are.
NICE and other HTA bodies emphasise clear decision problems, justified model structures, documented inputs, transparent assumptions, and sensitivity analyses. Medip applications make those elements visible before and after the model run.
Each application can define the population, intervention, comparator, outcomes, time horizon, perspective, and setting before results are interpreted.
Clinical effects, utilities, resource use, costs, and assumptions are documented and linked to literature, guidelines, databases, or expert input.
Application design, structural choices, health states, pathway events, extrapolations, and patient flow are available for review.
Analyses can separate base-case results from alternative assumptions, scenario analyses, and non-reference-case perspectives.
A documented application provides a reusable evidence asset for scientific communication, reimbursement planning, and local evidence adaptation.
Scientific publications
Documented application structure, model logic, parameter sources, uncertainty analysis, and outputs provide a basis for manuscripts, conference abstracts, and supplementary materials.
Value dossiers
The same application provides a structured evidence base for global value dossiers, local value messages, reimbursement discussions, and evidence gap planning.
HTA submissions
Inputs, assumptions, comparators, time horizons, perspectives, and sensitivity analyses are documented for technical review and country adaptation.
Stakeholder explanation
Technical users can inspect model logic while non-specialists can see why results change across patients, pathways, and assumptions selected in the application.
Users can see which inputs are evidence-based, which assumptions are local, which parameters drive uncertainty, and how changes propagate through costs, outcomes, QALYs, budget impact, and broader value.
Link application parameters to trials, literature, guidelines, registries, cost databases, and expert elicitation.
Document why the application uses a specific pathway, health-state structure, time horizon, and patient flow.
Identify which parameters drive outcomes and when the innovation becomes favourable.
Generate clear tables, figures, scenario outputs, and explanations for technical and non-technical review.

Join the discussion
We can walk through the application structure, model assumptions, and HTA evidence requirements for your technology.